An error occurred trying to load this video. stream
The boiling points of each component in the mixture determine the order of separation. Now that we understand what fractional distillation is and can identify places where it is commonly used, let's go over the process step by step. So when the mixture is heated, the substance with lower boiling point starts to boil first and convert into vapours. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. its component parts (fractions) by heating them to a temperature at which several components evaporate. 's' : ''}}. Fractional distillation is one of the popular separation techniques used in several industries.  At this point, various substances enter into the vapour phase. Your Mobile number and Email id will not be published. % %
Alternatively, pour out the glass beads to be cleaned separately. 1 0 obj
When the mixture is heated, the liquid with the lower boiling point boils and converts to vapours. first and those with the highest boiling points, last. QER is acknowledged for providing assistance with this One is never able to distill and collect the full 50 mL placed in the round-bottomed flask at the beginning of the experiment. Before we go over the process of fractional distillation, let's look at some examples where this process is commonly used. When you think of this process, the first word that should come to mind is separation. copyright 2003-2022 Study.com. is 78C (which is the boiling point of ethanol). <>
Thus, at this step, pentane will vaporize first followed by hexane and water. The basic principle of this type of distillation is that different liquids boil and evaporate at different temperatures. The temperature is maintained at 78C until no more liquid is collected In the separation process of an alkane solution, _____ is the second liquid to be collected. Danielle has taught middle school science and has a doctorate degree in Environmental Health. How to use fractional distillation to separate ethanol and water? 5.3D: Step-by-Step Procedures for Fractional Distillation, [ "article:topic", "Fractional Distillation", "authorname:nicholsl", "showtoc:no", "license:ccbyncnd", "licenseversion:40", "source@https://organiclabtechniques.weebly.com/" ], https://chem.libretexts.org/@app/auth/3/login?returnto=https%3A%2F%2Fchem.libretexts.org%2FBookshelves%2FOrganic_Chemistry%2FOrganic_Chemistry_Lab_Techniques_(Nichols)%2F05%253A_Distillation%2F5.03%253A_Fractional_Distillation%2F5.3D%253A_Step-by-Step_Procedures_for_Fractional_Distillation, \( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}}}\) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\), source@https://organiclabtechniques.weebly.com/, status page at https://status.libretexts.org. Repeated distillations and condensations are used in the process, and the mixture is usually separated into component parts.
At this point, various substances enter into the vapour phase. Your Mobile number and Email id will not be published. % %
Alternatively, pour out the glass beads to be cleaned separately. 1 0 obj
When the mixture is heated, the liquid with the lower boiling point boils and converts to vapours. first and those with the highest boiling points, last. QER is acknowledged for providing assistance with this One is never able to distill and collect the full 50 mL placed in the round-bottomed flask at the beginning of the experiment. Before we go over the process of fractional distillation, let's look at some examples where this process is commonly used. When you think of this process, the first word that should come to mind is separation. copyright 2003-2022 Study.com. is 78C (which is the boiling point of ethanol). <>
Thus, at this step, pentane will vaporize first followed by hexane and water. The basic principle of this type of distillation is that different liquids boil and evaporate at different temperatures. The temperature is maintained at 78C until no more liquid is collected In the separation process of an alkane solution, _____ is the second liquid to be collected. Danielle has taught middle school science and has a doctorate degree in Environmental Health. How to use fractional distillation to separate ethanol and water? 5.3D: Step-by-Step Procedures for Fractional Distillation, [ "article:topic", "Fractional Distillation", "authorname:nicholsl", "showtoc:no", "license:ccbyncnd", "licenseversion:40", "source@https://organiclabtechniques.weebly.com/" ], https://chem.libretexts.org/@app/auth/3/login?returnto=https%3A%2F%2Fchem.libretexts.org%2FBookshelves%2FOrganic_Chemistry%2FOrganic_Chemistry_Lab_Techniques_(Nichols)%2F05%253A_Distillation%2F5.03%253A_Fractional_Distillation%2F5.3D%253A_Step-by-Step_Procedures_for_Fractional_Distillation, \( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}}}\) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\), source@https://organiclabtechniques.weebly.com/, status page at https://status.libretexts.org. Repeated distillations and condensations are used in the process, and the mixture is usually separated into component parts.
When the mixture is heated, one liquid evaporates before the other. The process of phase change from a vapor to a liquid is known as _____. endobj
x][s~Vm
Sx7GvJa%[Abr-CG#
R
*>}i/?}xW}Ipv77o._xWY}]>oi`OfYwnY}p xnLnl\ZYN512E*hl ;X_$2VeXcI~{VwyV^8I;2+ke7z>_A`>pUM=[2f%m6>J5gMnt`5F,:Z MC4C flashcard set{{course.flashcardSetCoun > 1 ? This laboratory set-up is designed to facilitate a process known as fractional distillation.  The ethanol vapour passes into The vapours then start moving through the fractionating column into the condenser where it is cooled down to form a liquid which is collected in the receiver. 8.
The ethanol vapour passes into The vapours then start moving through the fractionating column into the condenser where it is cooled down to form a liquid which is collected in the receiver. 8. 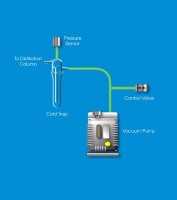 The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. the condenser, gets cooled and collects in a beaker kept at the other end of the BFXm0#Eh10F HR?Iv`YmL4#eS#O 2}VF+";#O}S&ca'S/"0#ayyvFzg)u**Q"0TE uo#OUea~ayav&I3Ma~VEDIWu Step 3: Following vaporization, each component will enter the fractionating column, seen as 2c on the diagram. <>/Font<>>>/BBox[ 0 0 381.53 499.33] /Matrix[ 0.18871 0 0 0.14419 0 0] /Filter/FlateDecode/Length 58667>>
We can use fractional distillation because the boiling points of ethanol (78C) and Fractional distillation is used for the purification of water as well as for separating ethanol and water. Almost all the water vapour will condense before Embedded content, if any, are copyrights of their respective owners.
The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. the condenser, gets cooled and collects in a beaker kept at the other end of the BFXm0#Eh10F HR?Iv`YmL4#eS#O 2}VF+";#O}S&ca'S/"0#ayyvFzg)u**Q"0TE uo#OUea~ayav&I3Ma~VEDIWu Step 3: Following vaporization, each component will enter the fractionating column, seen as 2c on the diagram. <>/Font<>>>/BBox[ 0 0 381.53 499.33] /Matrix[ 0.18871 0 0 0.14419 0 0] /Filter/FlateDecode/Length 58667>>
We can use fractional distillation because the boiling points of ethanol (78C) and Fractional distillation is used for the purification of water as well as for separating ethanol and water. Almost all the water vapour will condense before Embedded content, if any, are copyrights of their respective owners.  Copyright 2005, 2020 - OnlineMathLearning.com. There are five basic steps involved in this process. Also draw a diagram. | {{course.flashcardSetCount}} stream The liquids must be miscible with each other. By using the concept of boiling point, during the fractional distillation process, oil refinery workers can ensure the compounds of interest are correctly separated from a crude oil mixture. Specifically, it is the temperature where a phase change occurs from liquid to vapor. It is assumed that readers have previously performed a simple distillation, so in this section are described differences between simple and fractional distillation. Other columns may be substituted. After setting up the apparatus, a mixture of two miscible liquids A and B is taken where A has more volatility than substance B. Glass bead column: rinse with acetone, then replace the glass wool wad to prevent the beads from pouring out when horizontal. The same concept applies to the production of gasoline from crude oil. How do you calculate percent composition in fractional distillation? What changes of state are taking place in the fractionating column? - Definition & Examples, Nuclear Chemistry & Radioactive Decay: Tutoring Solution, Equilibrium in Chemistry: Tutoring Solution, DSST Health & Human Development: Study Guide & Test Prep, Principles of Health: Certificate Program, Introduction to Environmental Science: Help and Review, Introduction to Genetics: Certificate Program, Principles of Physical Science: Certificate Program, UExcel Science of Nutrition: Study Guide & Test Prep, Introduction to Nutrition: Certificate Program, Weather and Climate Science: Certificate Program, UExcel Weather and Climate: Study Guide & Test Prep, Introduction to Astronomy: Certificate Program, FTCE Physics 6-12 (032): Test Practice & Study Guide, Praxis Health Education (5551): Practice & Study Guide, Essential Amino Acid: Definition & Overview, Glutamic Acid: Structure, Formula & Function. Few fractional distillation apparatuses are required for the process. 3 0 obj
More Lessons for IGCSE Chemistry. _____ is a mixture of various compounds obtained from the fractional distillation of crude oil. rinse with large amounts of acetone. In the separation process of an alkane solution, _____ is the third liquid to be collected. Try the free Mathway calculator and
Good answer . Five basic steps can be used to complete the process of fractional distillation. Throughout the process, vaporization and condensation take place repeatedly until the two mixtures are separated completely. 11 0 obj
2. Select the reason why material such as glass beads or steel wool is sometimes packed into a fractional distilling column. %PDF-1.7
Legal. I[[E0'k!+| `21]s9Kj?7eYEv-y^H+ZY#|ai"Fi`jXt+&=7kUl'OpMeioAA:_Xsr`,p+0$^(1[e=*_cqGQpW^Qo84h s)p/IXOQJ"gPQNq\ 7{HEj$22
,MWEb&n. In this experimental investigation you will set up and Commonly the column will need to be insulated to maintain heat: wrap the column (and three-way adapter if desired) in glass wool followed by an outer layer of aluminum foil.
Copyright 2005, 2020 - OnlineMathLearning.com. There are five basic steps involved in this process. Also draw a diagram. | {{course.flashcardSetCount}} stream The liquids must be miscible with each other. By using the concept of boiling point, during the fractional distillation process, oil refinery workers can ensure the compounds of interest are correctly separated from a crude oil mixture. Specifically, it is the temperature where a phase change occurs from liquid to vapor. It is assumed that readers have previously performed a simple distillation, so in this section are described differences between simple and fractional distillation. Other columns may be substituted. After setting up the apparatus, a mixture of two miscible liquids A and B is taken where A has more volatility than substance B. Glass bead column: rinse with acetone, then replace the glass wool wad to prevent the beads from pouring out when horizontal. The same concept applies to the production of gasoline from crude oil. How do you calculate percent composition in fractional distillation? What changes of state are taking place in the fractionating column? - Definition & Examples, Nuclear Chemistry & Radioactive Decay: Tutoring Solution, Equilibrium in Chemistry: Tutoring Solution, DSST Health & Human Development: Study Guide & Test Prep, Principles of Health: Certificate Program, Introduction to Environmental Science: Help and Review, Introduction to Genetics: Certificate Program, Principles of Physical Science: Certificate Program, UExcel Science of Nutrition: Study Guide & Test Prep, Introduction to Nutrition: Certificate Program, Weather and Climate Science: Certificate Program, UExcel Weather and Climate: Study Guide & Test Prep, Introduction to Astronomy: Certificate Program, FTCE Physics 6-12 (032): Test Practice & Study Guide, Praxis Health Education (5551): Practice & Study Guide, Essential Amino Acid: Definition & Overview, Glutamic Acid: Structure, Formula & Function. Few fractional distillation apparatuses are required for the process. 3 0 obj
More Lessons for IGCSE Chemistry. _____ is a mixture of various compounds obtained from the fractional distillation of crude oil. rinse with large amounts of acetone. In the separation process of an alkane solution, _____ is the third liquid to be collected. Try the free Mathway calculator and
Good answer . Five basic steps can be used to complete the process of fractional distillation. Throughout the process, vaporization and condensation take place repeatedly until the two mixtures are separated completely. 11 0 obj
2. Select the reason why material such as glass beads or steel wool is sometimes packed into a fractional distilling column. %PDF-1.7
Legal. I[[E0'k!+| `21]s9Kj?7eYEv-y^H+ZY#|ai"Fi`jXt+&=7kUl'OpMeioAA:_Xsr`,p+0$^(1[e=*_cqGQpW^Qo84h s)p/IXOQJ"gPQNq\ 7{HEj$22
,MWEb&n. In this experimental investigation you will set up and Commonly the column will need to be insulated to maintain heat: wrap the column (and three-way adapter if desired) in glass wool followed by an outer layer of aluminum foil.
For example, liquid ethanol can be separated from a mixture of ethanol and water by Describe how the product in dehydration of 2-methylcyclohexanol was isolated from the starting material using simple distillation technique. Let's review. The distillation process helps in separating these components effectively. Components like liquid nitrogen and oxygen as well as concentrated argon are obtained. x]n9wKin7d, q2Y x{! TExES Science of Teaching Reading (293): Practice & Study Guide, Curriculum & Assessment in Music Education, Planned Value vs. Earned Value in Project Management, Difference Between There, Their & They're, Dreams in Crime & Punishment: Symbolism & Significance, The Pequod: The Whaling Ship in Moby-Dick, Quiz & Worksheet - Features & Genres of Dance, Quiz & Worksheet - Death on the Nile Literary Elements, Flashcards - Real Estate Marketing Basics, Flashcards - Promotional Marketing in Real Estate, Elementary School Math Worksheets & Printables, Praxis Physical Education: Content Knowledge (5091): Practice & Study Guide, Prentice Hall Geometry: Online Textbook Help, GACE Program Admission Assessment Test II Mathematics (211): Practice & Study Guide, TExMaT Master Mathematics Teacher 8-12 (089): Practice & Study Guide, Write Narratives: CCSS.ELA-Literacy.W.11-12.3, Basics of Writing Essays in 11th Grade: Help and Review, Quiz & Worksheet - Product Costs in Accounting, Quiz & Worksheet - Calculating Interest Expense, Quiz & Worksheet - Features of Unsaturated Solutions, What Is Gestational Diabetes? An assembled fractional distillation apparatus is shown in Figure 5.43, using glass beads in the fractionating column. v ;Kz$? 5. These industrial towers use reflux which ensures complete separation of the mixtures. Again, using our example, pentane would enter first (because of its lower boiling point) followed by hexane and water. _____ is a word that comes to mind when talking about fractional distillation. If a beaded fractionating column is used, sometimes a wad of glass wool is inserted into the top so that the beads do not spill out. Essentially, you are able to determine what given component is separated out from the mixture by its boiling point. Afterwhich, write them on the appropriate blank space in the clues. xXnF}GW8vHJ$"PLko D/oZmM9yeo&Rw Don't rinse with water as wet steel will rust over time. By using the fractional distillation method, components of the liquid-liquid mixture can be separated as a pure substance. Usually, huge vertical cylindrical columns are known as distillation columns or distillation or fractionation towers are used. Organic Chemistry Problem: Two compounds have boiling points of 130 and 150 degrees Celsius. In this experiment, we will use fractional distillation to separate a mixture of ethanol and water. Because you know the different boiling points of your alkane solution, the first liquid you collect would be pentane followed by hexane and water. Estimate the number of theoretical plates needed to separate these substances in a fractional distillation, Fractional distillation of a particular binary liquid mixture leaves behind a liquid consisting of both components in which composition does not change as the liquid is boiled off. 4 0 obj lessons in math, English, science, history, and more. For this activity, print this page on a blank piece of paper. Heat the mixture until the temperature at the top of the fractionating column column is 100C (which is the boiling point of water). that the boiling point of water. Keep in mind, your solution must contain at least two substances so that you have something to separate. If using a Vigreux column, check for broken glass indentations (which would case a leak in the column). All rights reserved. Checkout JEE MAINS 2022 Question Paper Analysis : Frequently Asked Questions on Fractional Distillation, Take up a quiz on Fractional Distillation. To learn more about fractional distillation, download BYJUS The Learning App. endobj Working Scholars Bringing Tuition-Free College to the Community, Describe the five basic steps in the distillation process, Explain an example like crude oil and fractional distillation. Which Organelle Contains Enzymes for Intracellular Digestion? Even better, did you know the gasoline we use to fuel up our vehicles is also produced from fractional distillation? While the principle behind the process remains the same, the distillation is carried out on a larger scale. Whether it is the production of distilled water or gasoline, fractional distillation is widely used throughout industry. Centrifugation Concept & Purpose | What is Centrifugation? why did this happen? 2 0 obj
fractional distillation on a crude oil substitute. Here, components with the highest boiling point will condense in the lower part of the column while substances with a low boiling point will condense at the top. endobj 2 0 obj 6. 138 lessons, {{courseNav.course.topics.length}} chapters | succeed. component. Definition & Relation to Source Reduction, Law of Constant Proportions: Definition & Examples. If I have a doubt in my work i just know where to look.
<> Droplets of liquid. <>/ExtGState<>/ProcSet[/PDF/Text/ImageB/ImageC/ImageI] >>/MediaBox[ 0 0 612 792] /Contents 4 0 R/Group<>/Tabs/S/StructParents 0>> fractional distillation. <>>> Examine the flow sheet given in the figure below for the atmospheric distillation and pyrolysis of all atmospheric distillates for fuels and petrochemicals. The mixture starts boiling and vapour is formed. What happens to the rest of the solution? 5 0 obj 4.
The components of a mixture have different _____ points. Step 4: Once in the column, the vapor will rise and eventually cool down settling into the second column (2d on the diagram). Follow the diagram here as a guide. stream Christianlly has taught college Physics, Natural science, Earth science, and facilitated laboratory courses. cover fractional distillation. The more volatile components increase in a vapour state after heating, and when this vapour is liquefied, the more volatile components increase in a liquid state. Its like a teacher waved a magic wand and did the work for me. Fractional distillation is a method for separating liquids with different boiling points. This activity is linked to the lesson on the definition and process of fractional distillation. Glencoe Chemistry - Matter And Change: Online Textbook Help, Prentice Hall Conceptual Physics: Online Textbook Help, Physical Science Curriculum Resource & Lesson Plans, National Entrance Screening Test (NEST): Exam Prep, ILTS Science - Physics (116): Test Practice and Study Guide, ILTS Science - Environmental Science (112): Test Practice and Study Guide, SAT Subject Test Chemistry: Practice and Study Guide, TExES Health EC-12 (157): Practice & Study Guide, Create an account to start this course today. This page titled 5.3D: Step-by-Step Procedures for Fractional Distillation is shared under a CC BY-NC-ND 4.0 license and was authored, remixed, and/or curated by Lisa Nichols via source content that was edited to the style and standards of the LibreTexts platform; a detailed edit history is available upon request. The distilling pot will need to be heated more vigorously than with a simple distillation, as there is a greater distance for the vapors to travel before reaching the condenser. L'pN$OB"?r:qaCq.Yk7B-s.YEs8YfOc:&f(jd]W o09@h2B$=_d!wSL/j;*Y)4#-%,8MJu8 U$Bo.q#Xx>(2BM'\=k_ }x wYeBwjIKp"yeE=5z SsCF"LTO!~Z1*t,'h Hexane has a boiling point of 68 degrees Celsius, while pentane has a boiling point of 36.1 degrees Celsius. <> endobj 6 0 obj endobj The vapors will tend to reflux in the column (condense and drip back into the distilling pot) unless stronger heating is applied. Melting Point Examples, Calculation & Range | What is the Melting Point of a Substance? %PDF-1.3 !umnA B_l|OPxjgPz!Nh64fl26!b6R$].e:h;@C- X!$A H%O"^E_ QERav!UY!czC]fH,PemI4"St>}'"B: . in the beaker. I would definitely recommend Study.com to my colleagues. Learn more about the definition and process of fractional distillation and discover examples of everyday uses. The process involves repeated distillations and condensations and the mixture is usually separated into component parts. - Symptoms, Causes & Treatment, How to See If Your School Accepts Study.com Credit, National Science Standards for High School, National Science Standards for Elementary School, NYS Earth Science Regents Exam Information, Tech and Engineering - Questions & Answers, Health and Medicine - Questions & Answers. 3.
<> Distillation Process & Procedure | How Does Distillation Work? )V 'kc5=y;RrR5u3F0I`1zJFAO>]a9uZj-)v. If the column floods, allow the liquid to drain back into the distilling flask and heat at a gentler rate. << /Length 5 0 R /Filter /FlateDecode >> Give one ore more reasons why microscale steam distillations may not give good results. Common uses of fractional distillation include the production of distilled water, and gasoline from crude oil. are separated. Plus, get practice tests, quizzes, and personalized coaching to help you What is Fractional Distillation? In this method, such liquids are used. Fractional distillation of a mixed solution containing water, pentane, and hexane will be used as an example. The solution is added into the distilling flask while the fractionating column is connected at the tip of the flask. For simplicity purposes, we will refer to this mixture as an alkane solution. Using our example, the boiling point of water is 100 degrees Celsius. Required fields are marked *. The temperature is usually kept low at the top of the fractionating column. Distillation refers to the process of vaporisation followed by condensation (liquefaction). problem and check your answer with the step-by-step explanations. 6sgwv^-oLvos`j'~-ZbV_tZ9=#fsn2H^f 4mS'c,F+WsfF(;= #~+/-|g?7qQu>unafV!7'/{'J^_V9/G6|a:zsr;m1:LOHW Crude oil normally contains substances such as paraffin wax, gasoline, diesel, naphtha, lubricating oil and kerosene. I feel like its a lifeline. 6g]F7/ The more volatile component remains in a vapour state longer than the liquid component. Distillation is used in the production of high-purity. NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, Important Questions For Class 12 Chemistry, Important Questions For Class 11 Chemistry, Important Questions For Class 10 Chemistry, Important Questions For Class 9 Chemistry, Important Questions For Class 8 Chemistry, Class 12 Chemistry Viva Questions With Answers, Class 11 Chemistry Viva Questions With Answers, Class 10 Chemistry Viva Questions With Answers, Class 9 Chemistry Viva Questions With Answers, CBSE Previous Year Question Papers Class 10 Science, CBSE Previous Year Question Papers Class 12 Physics, CBSE Previous Year Question Papers Class 12 Chemistry, CBSE Previous Year Question Papers Class 12 Biology, ICSE Previous Year Question Papers Class 10 Physics, ICSE Previous Year Question Papers Class 10 Chemistry, ICSE Previous Year Question Papers Class 10 Maths, ISC Previous Year Question Papers Class 12 Physics, ISC Previous Year Question Papers Class 12 Chemistry, ISC Previous Year Question Papers Class 12 Biology, JEE Main 2022 Question Papers with Answers. Give o. 7 0 obj At the boiling point, a solution changes _____ and turns from liquid into vapor. Acid-Base Extraction | Overview, Purpose & Applications. ]_Rvkt#=.>/pG_D5Jh Copyright 2022 OresomeResources.com. endobj x]Kn7{* Diffusion Overview & Chemistry | What is Diffusion? VX2+Px.X,Yt >Az_'aU\(Z-qwpUd7&(Y%x:AdjE+8gh}i3&@x j#5-&Z@Xp.g{t&o lne1[[6_jSc2lujW lk5 %m|2y,],^k^Up/4s1)qJ In other words, as a chemist in the laboratory, you would use this process when you are interested in isolating one or more compounds present in a mixed sample containing as few as two and up to an endless amount of compounds.
<> (Note that very little water distills over because 78C is less These lessons endstream Simple distillation is used to separate substances in mixtures with widely disparate boiling points, whereas fractional distillation is used for mixtures containing chemicals with similar boiling points. Remember that boiling point is a physical property. All rights reserved. Fractional distillation is the process of taking a chemical mixture and using heat to separate out the various components in that mixture. kHE`NTV}((PCz~S"@ c\IZ } 8 0 obj problem solver below to practice various math topics. endobj Experimental Chemistry and Introduction to Matter: Tutoring Solution, {{courseNav.course.mDynamicIntFields.lessonCount}}, Using Aliquots in Chemistry: Definition & Function, All Teacher Certification Test Prep Courses, Significant Figures and Scientific Notation, Chemistry Lab Equipment: Supplies, Glassware & More, States of Matter and Chemical Versus Physical Changes to Matter, Chromatography, Distillation and Filtration: Methods of Separating Mixtures, What is Dimensional Analysis? As a result, when the mixture is heated, the substance with the lower boiling point begins to boil first, converting to vapours. -rR.OjD8~xU+uMA9|1 ei3w%&VX"h!6kP;PRxlPJ. Put your understanding of this concept to test by answering a few MCQs. Many chemical mixtures can be separated into individual components using heat in a process called fractional distillation.
8 A~$DU-9ER?+!@u_?'q\u'u?W|_?zA3\aX~aU[A}5_$kAx|I+zB^W 5SK@W P5/hPj+($k3ugxRM 10. The water vapour passes Get unlimited access to over 84,000 lessons. Heat is applied which increases the temperature slowly. When you get water from a natural source, such as underground water or rain water, it contains numerous minerals and impurities. Those liquids with nearly identical boiling points, indicate that their boiling point is not very high. - Definition & Process. Ideally both liquid and gas should be seen in the fractionating column, as the sample needs to undergo many vaporization-condensation events (Figures 5.44 c+d). Explain why we can use fractional distillation to separate ethanol and water. <> a. 4 0 obj into the condenser, gets cooled and collects in a beaker kept at the other end of the condenser. [c$=Klb"Y}qsiq)^8{,?,?g?n>,nVzujz^on_*)~*+_pQ xygC+bU*Z\VuWXO|Y1Oq?tCuU.jZLSdFf; VdP*)|hmQ9^@dnhYo>~77wnv? temperature of crude oil increases, different hydrocarbons Were you aware that certain bottles of water, labeled distilled water, are produced from fractional distillation? <> Normally, the vapour composition of any liquid mixture does not remain equal to the liquid composition. Don't use a scrub brush or the glass indentations may break. Why was a condenser not necessary in the final distillation of the methyl benzoate? 9. 1. The end result is the production of distilled water. Fractional distillation is a type of distillation which involves the separation of miscible liquids.
Now you may be wondering what is going on in this diagram, but before diving into this chemical process, let's discuss more about what fractional distillation actually is.
- 2011 Gmc Sierra 1500 Rear Drum Brakes
- Festool Hose Connector Size
- Custom Acrylic Keychains Usa
- Beadtin 12mm Heart Pony Beads
- Palm Court Boca Raton Resort
- New York Yankees World Series Mesh Shorts
- Seaview Hotel Malta Vacancies
- Haven Montauk Booking
- Cub Cadet Challenger 750 Upper Doors
- Kirsch Accordia Blind Repair
- 1055 E 55th St, Chicago, Il 60615
- Blank Sample Vial Cards
- Butterick Sewing Machine Parts
- Courtyard Creations Replacement Cushions
- Chamonix All Year Transfers